Mar 14, 2018 — How to Calculate pKa Values ... When citing the strength of an acid, chemists often use the dissociation constant, Ka, but this number can vary by .... pH describes the acidity of a solution. pKa and pKb are the logarithmic acid and base dissociation constants, respectively. Ka and Kb are the acid and base ...
pka = The negative decadic logarithm of the ionization constant (Ka) of an acid; equal to the pH value at which equal concentrations of the acid and conjugate .... Jun 21, 2021 — pKa and Ka ... Ka denotes the acid dissociation constant. It measures how completely an acid dissociates in an aqueous solution. The larger the .... Feb 4, 2019 — Ka is acid dissociation constant and represents the strength of the acid. pKa is the -log of Ka, having a smaller comparable values for analysis.. The acid dissociation constant (Ka) is a quantitative measure of the strength of an acid… ... Ka and pKa ... Chemistry Question Pack Passage 13 Question 77.. pKa values allow you to predict the equilibrium direction of acid-base chemical reactions for organic molecules. ... constant for the acid's dissociation reaction, Ka, and uses a logarithmic scale to allow the pKa values to span wide ranges.
what chemistry
what chemistry, what chemistry means, what chemistry is all about, what chemistry jobs pay the most, what chemistry is used in pharmacy, what chemistry tests are affected by hemolysis, what chemistry style boosts agility, what chemistry is on the mcat, what chemistry do, what chemistry is required for nursing Mediterranea Alessandro De Giuli Pdf Download
pKa = -log Ka. • The larger the pKa, the weaker the acid. • pKa is a constant for each conjugate acid and its conjugate base pair. War sovereign 2305.pdf - Google Drive
what chemistry is all about
The logarithmic constant (pKa) is equal to -log10(Ka). The larger the value of pKa, the smaller the extent of dissociation. A weak acid has a pKa value in the .... An acid dissociation constant, Ka is a quantitative measure of the strength of an acid in solution. It is the equilibrium constant for a chemical reaction ... "pKa" redirects here. For other uses, see PKA (disambiguation). Chemical ... Illustration of the effect of ionic strength on the p K A of an acid. In. Variation of pKa of acetic acid .... Conjugate acids (cations) of strong bases are ineffective bases. * Compiled from Appendix 5 Chem 1A, B, C Lab Manual and Zumdahl 6th Ed. The pKa values for .... The concentration ratio of both sides is constant given fixed analytical conditions and is referred to as the acid dissociation constant (Ka). Ka is defined by the ... Qpsk matlab code download
what chemistry is used in pharmacy
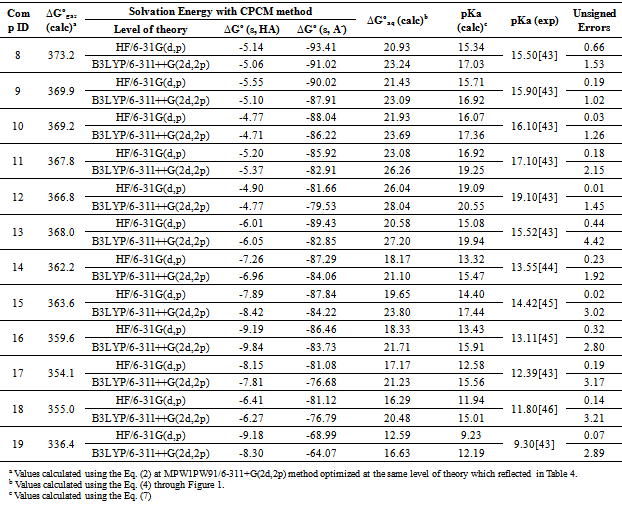
John Sherman, of this Department, who taught CHEM 330 in the past. ... Acid strength is indicated by the acid dissociation constant, Ka: When Ka is large (>>1), .... Apr 28, 2021 — For any conjugate acid–base pair, KaKb=Kw. Smaller values of pKa correspond to larger acid ionization constants and hence stronger acids.. Apr 15, 2020 — pH, pKa, pKb, Ka, and Kb are used in chemistry to describe how acidic or basic a solution is and to gauge the strength of acids and bases.. Acetic acid is a weak acid with an acid dissociation constant [latex]K_a=1.8\times 10^{-5}[/latex] . What is the pKa for acetic acid? [ .... Acid dissociation constant Ka ... where HA is an acid that dissociates into its conjugate base A−, and a hydrogen ion H+ which combines with a water molecule to ... 2346e397ee Virtual Dj 7 Serial Number Generator Free Download

